Lab Result: MOTS-c 10 mg (Batch CS-mc10-0309)
- Purity
- 99.443%
- Mass
- 11.89 mg
- Result Date
- March 18, 2026
- Date Added
- May 6, 2026
 Click to open report
Click to open report
Lumera Labs publishes the third-party Certificate of Analysis for every lot we ship. Each report shows reverse-phase HPLC purity, LC-MS identity confirmation, and LAL endotoxin clearance — matched to the batch number printed on your vial.
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
 Click to open report
Click to open report
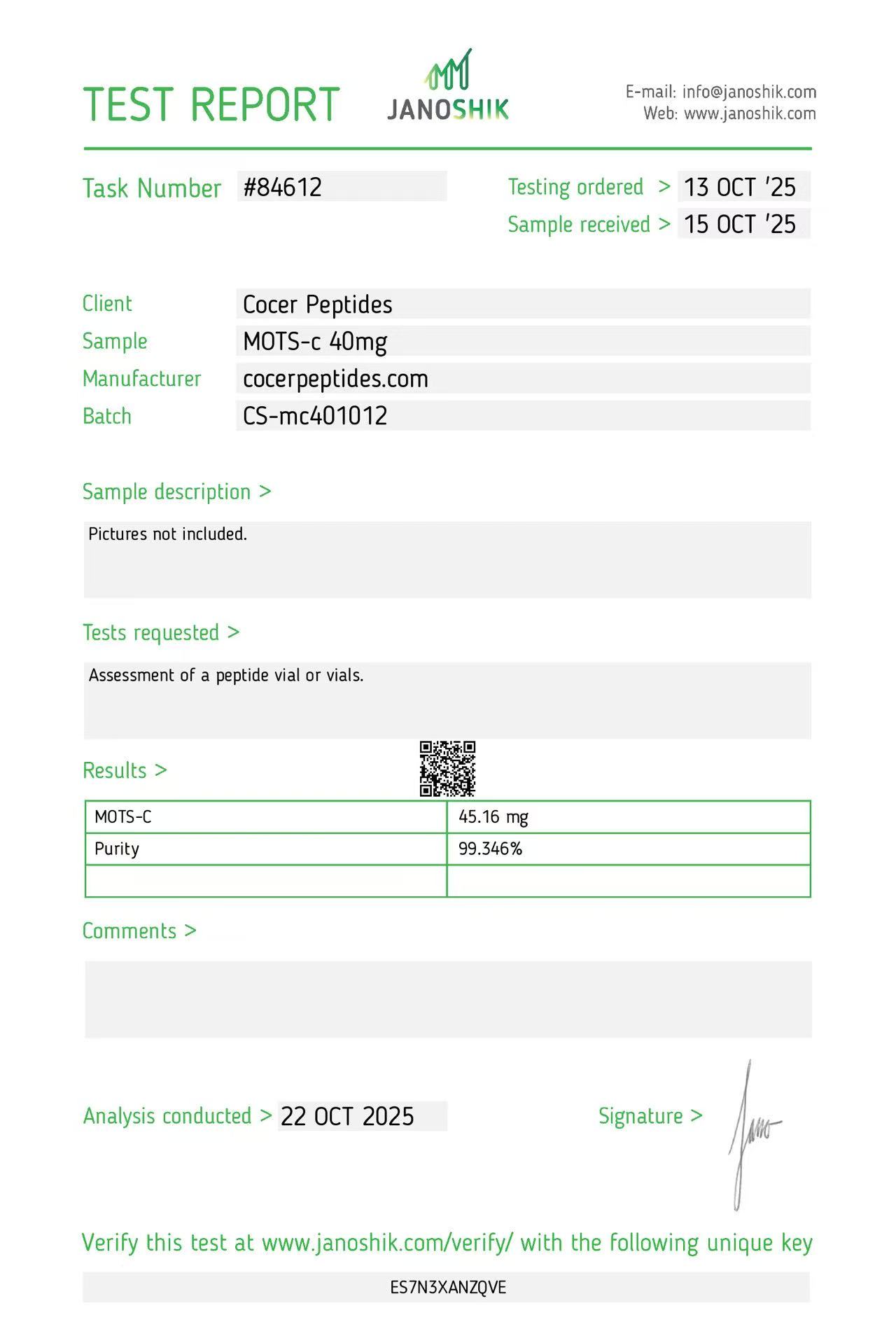 Click to open report
Click to open report
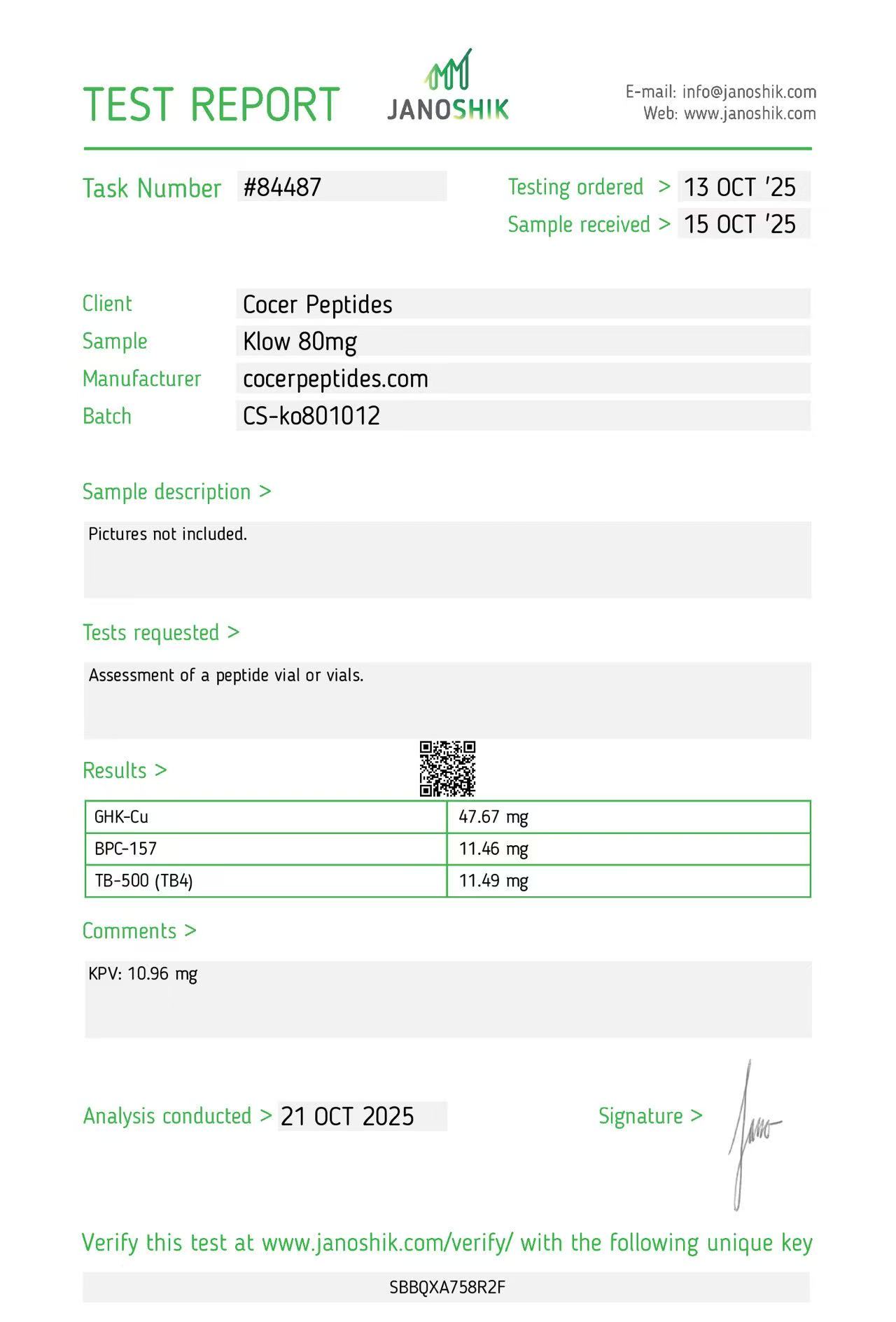 Click to open report
Click to open report
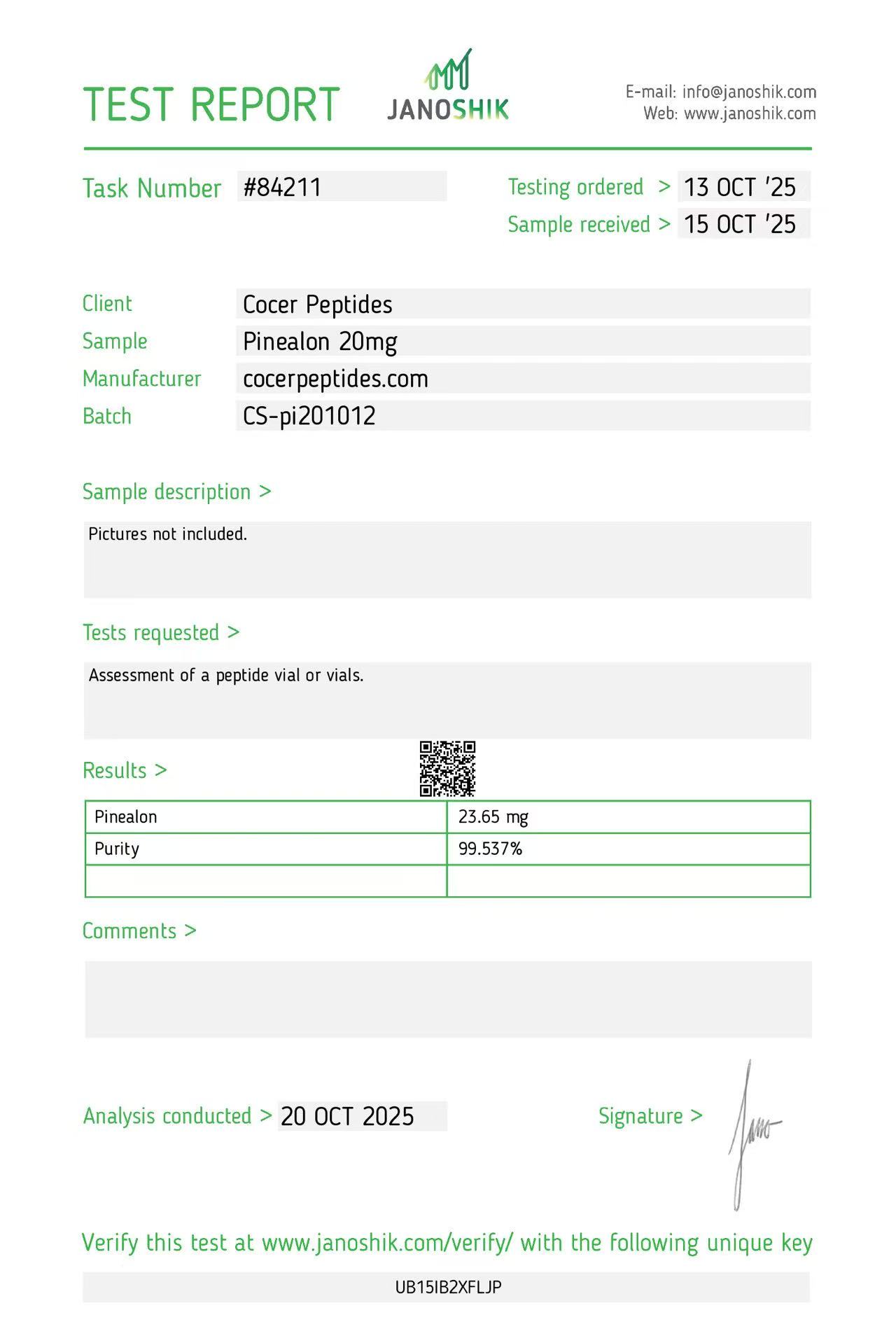 Click to open report
Click to open report
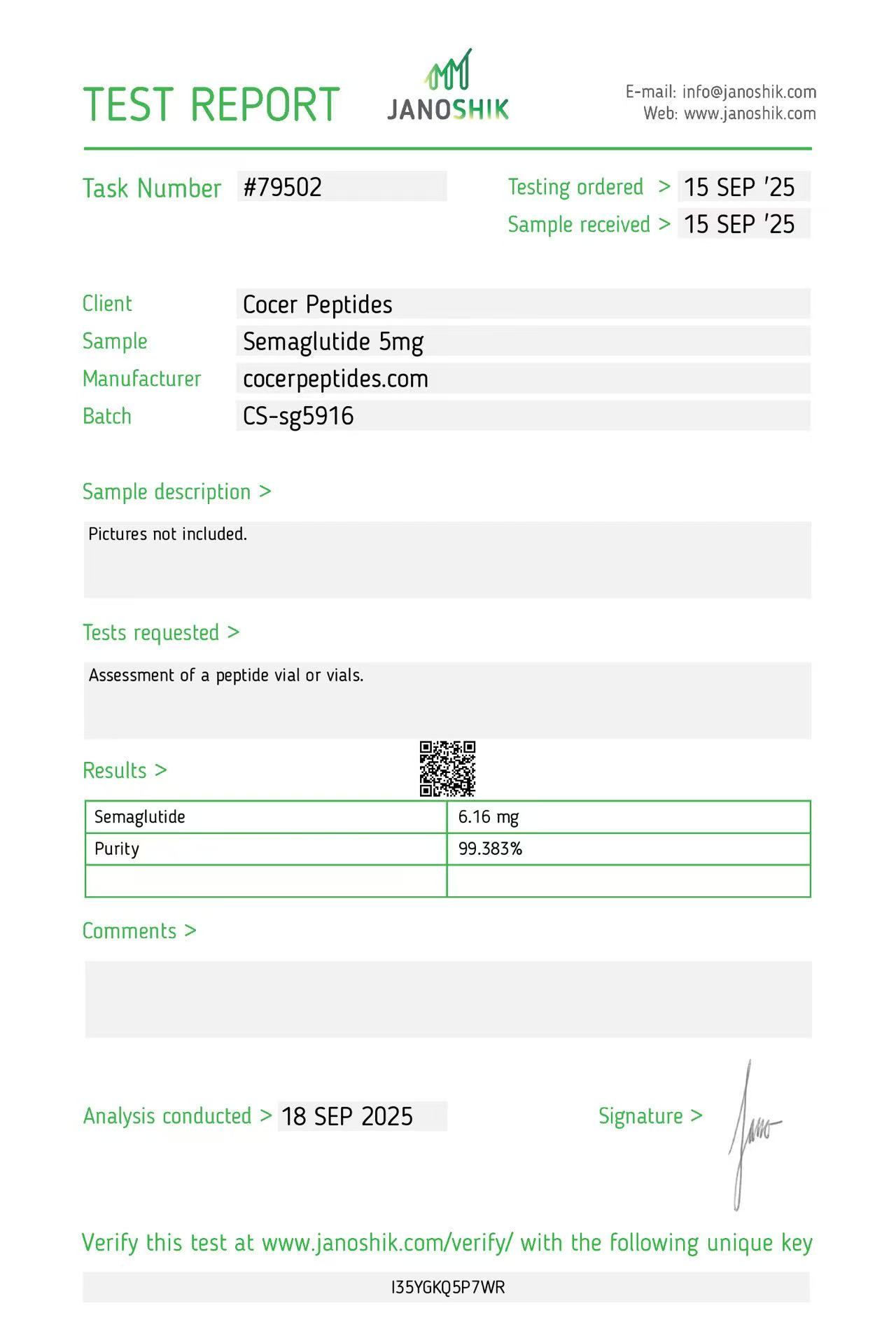 Click to open report
Click to open report
 Click to open report
Click to open report
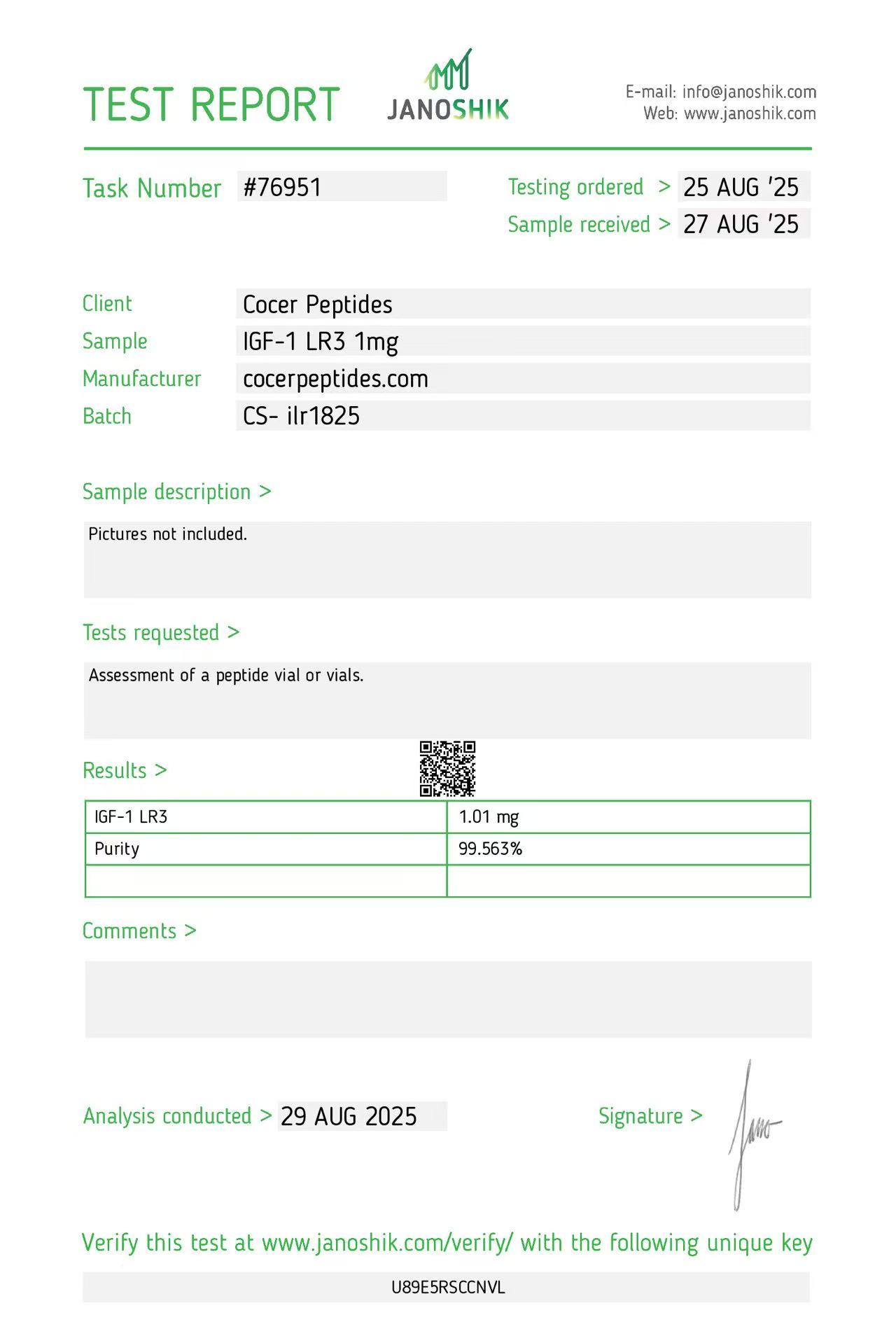 Click to open report
Click to open report
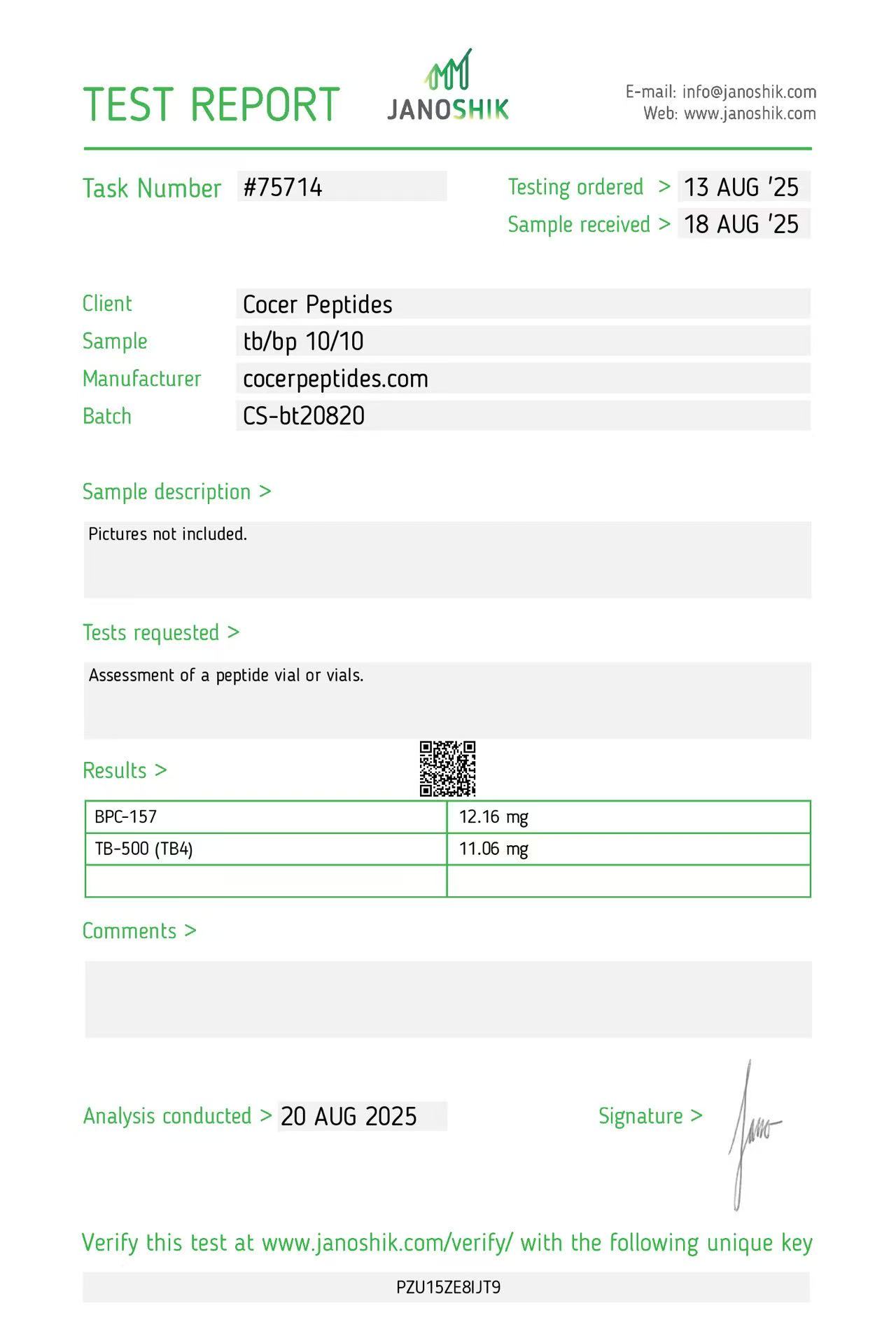 Click to open report
Click to open report
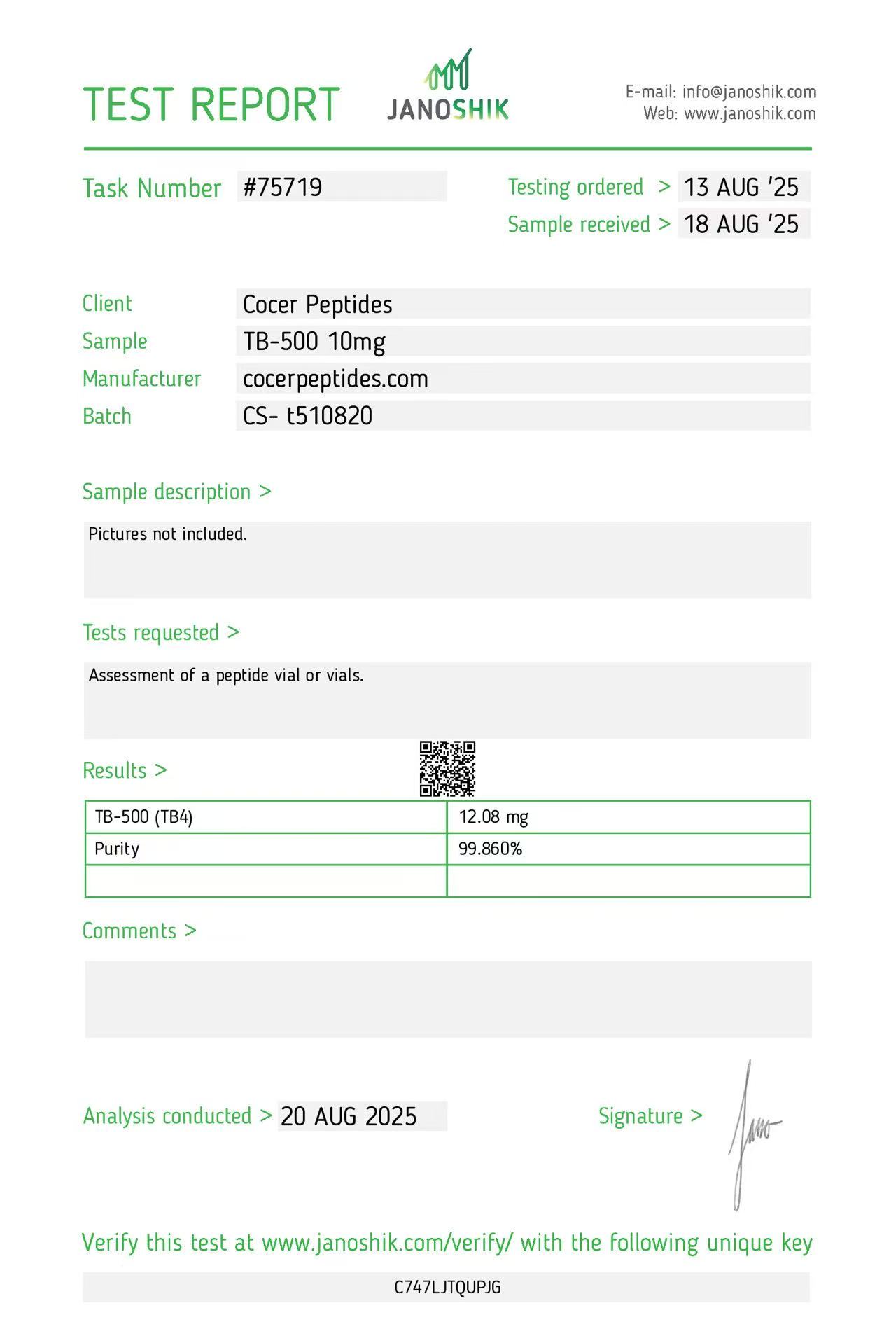 Click to open report
Click to open report
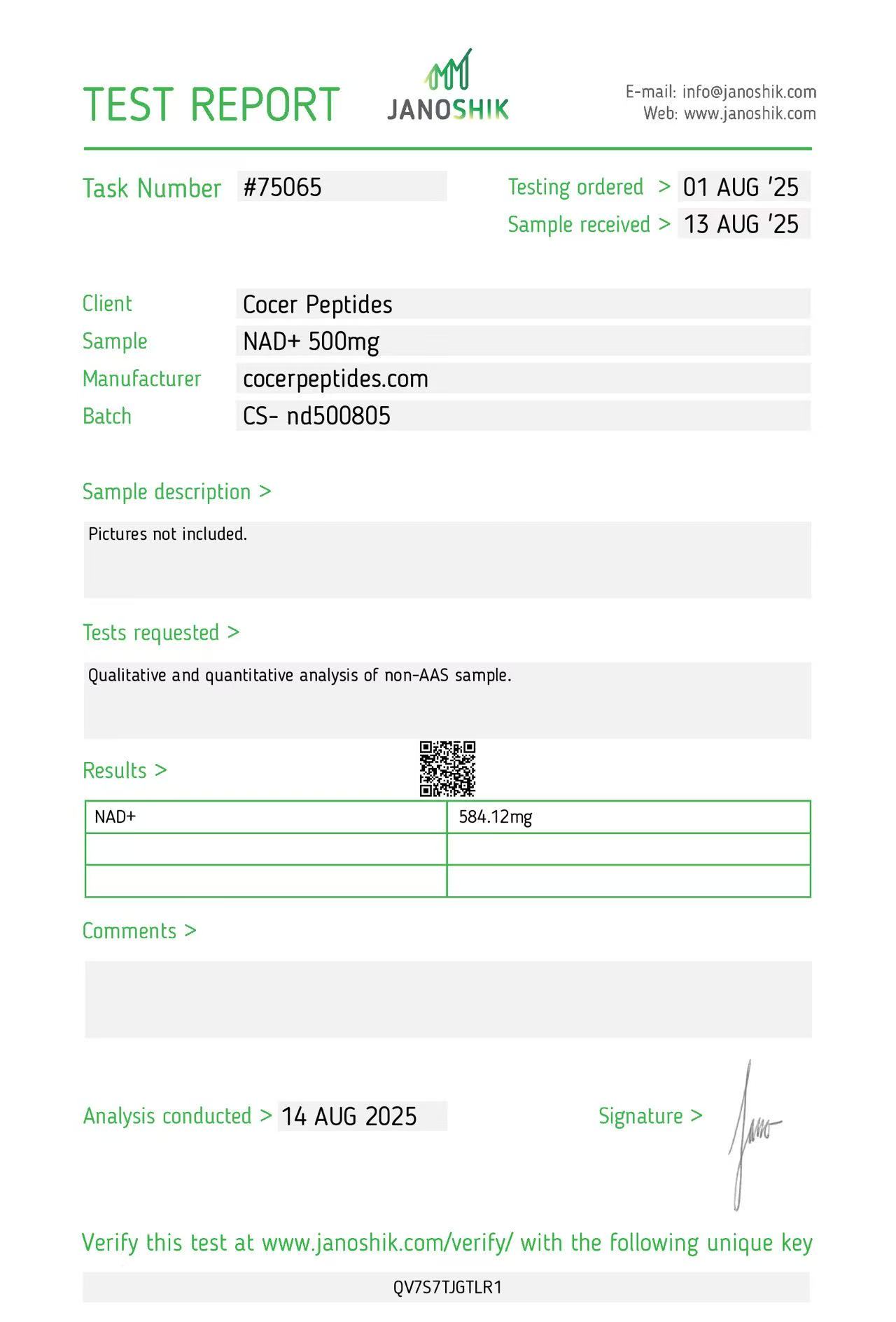 Click to open report
Click to open report
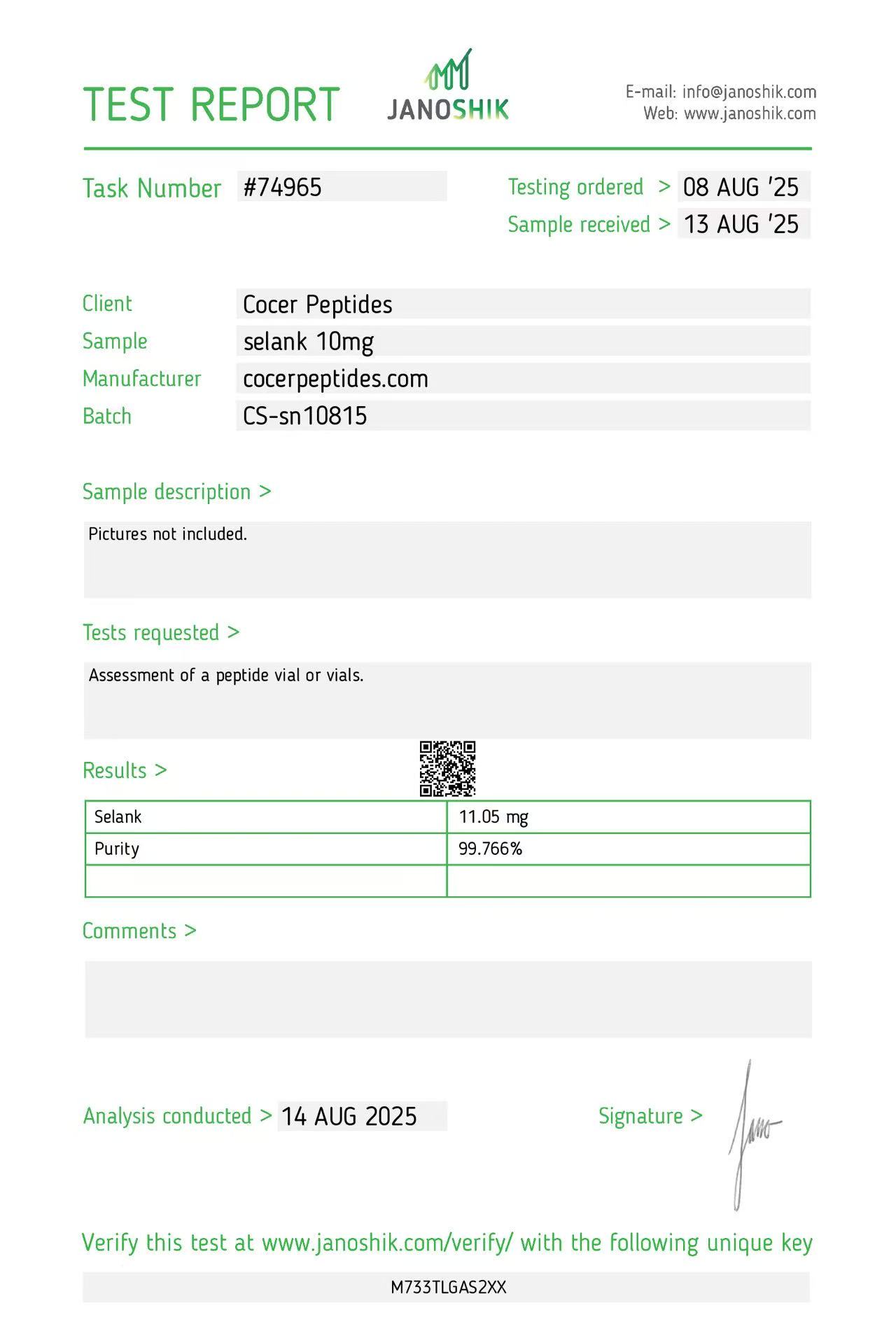 Click to open report
Click to open report
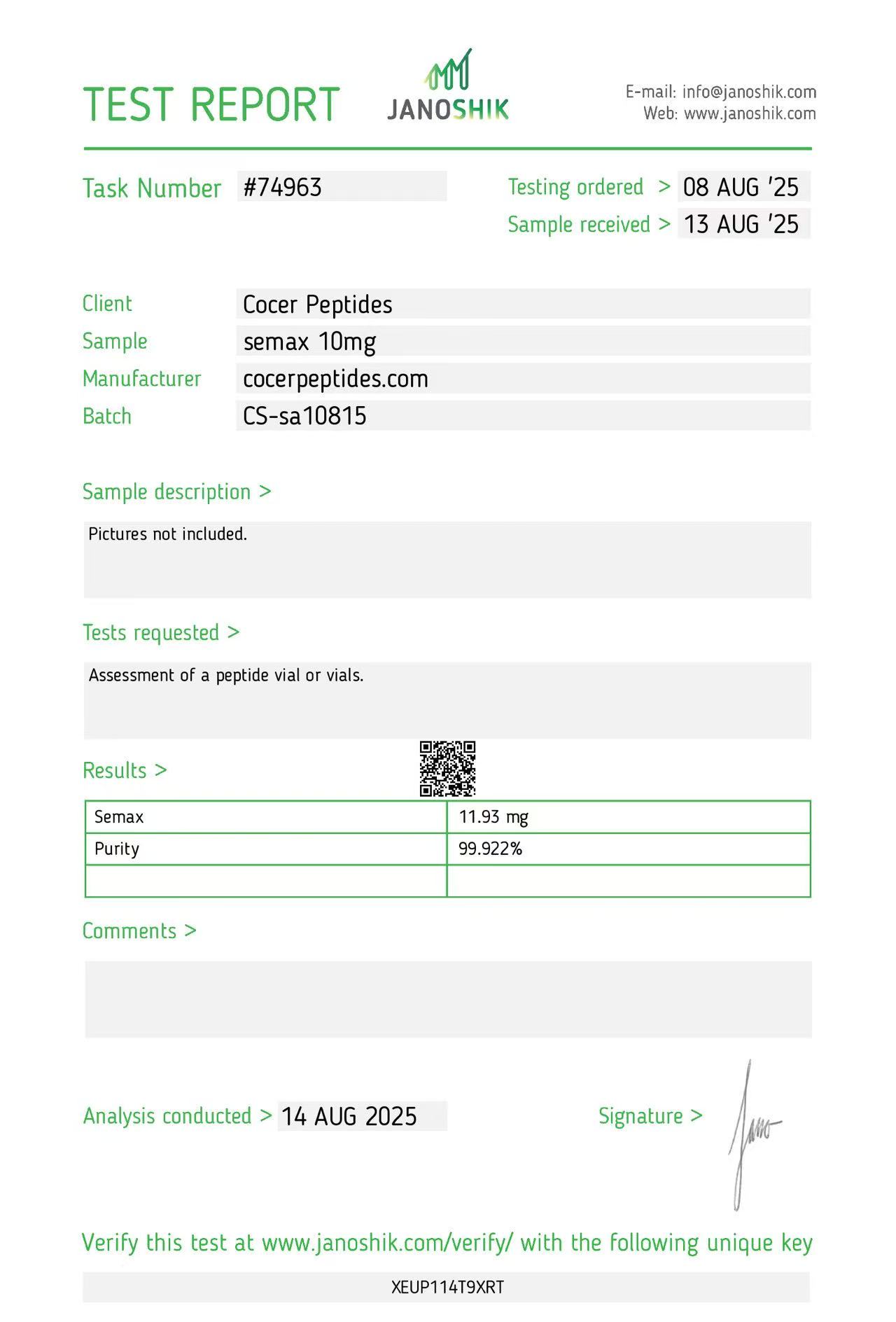 Click to open report
Click to open report
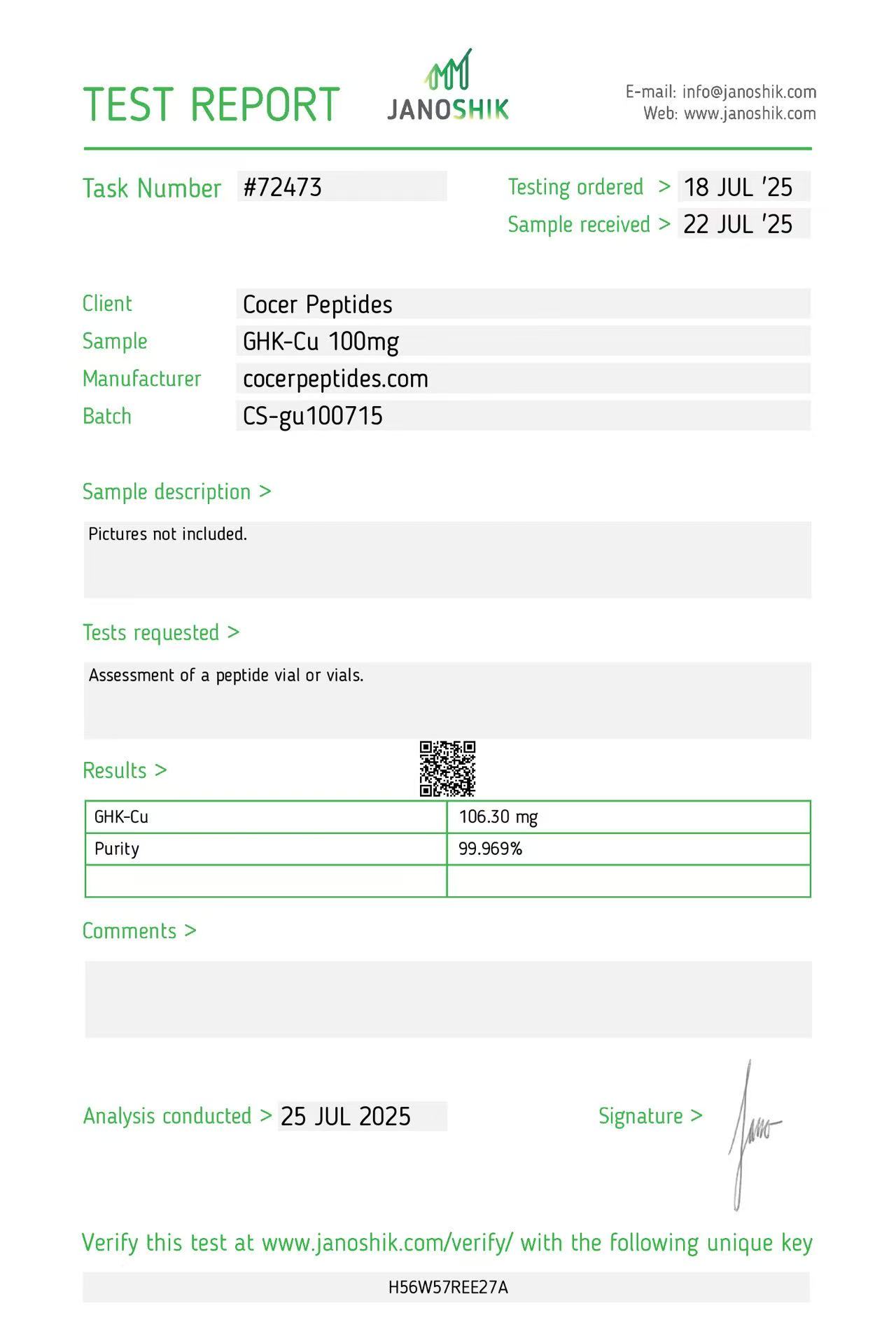 Click to open report
Click to open report
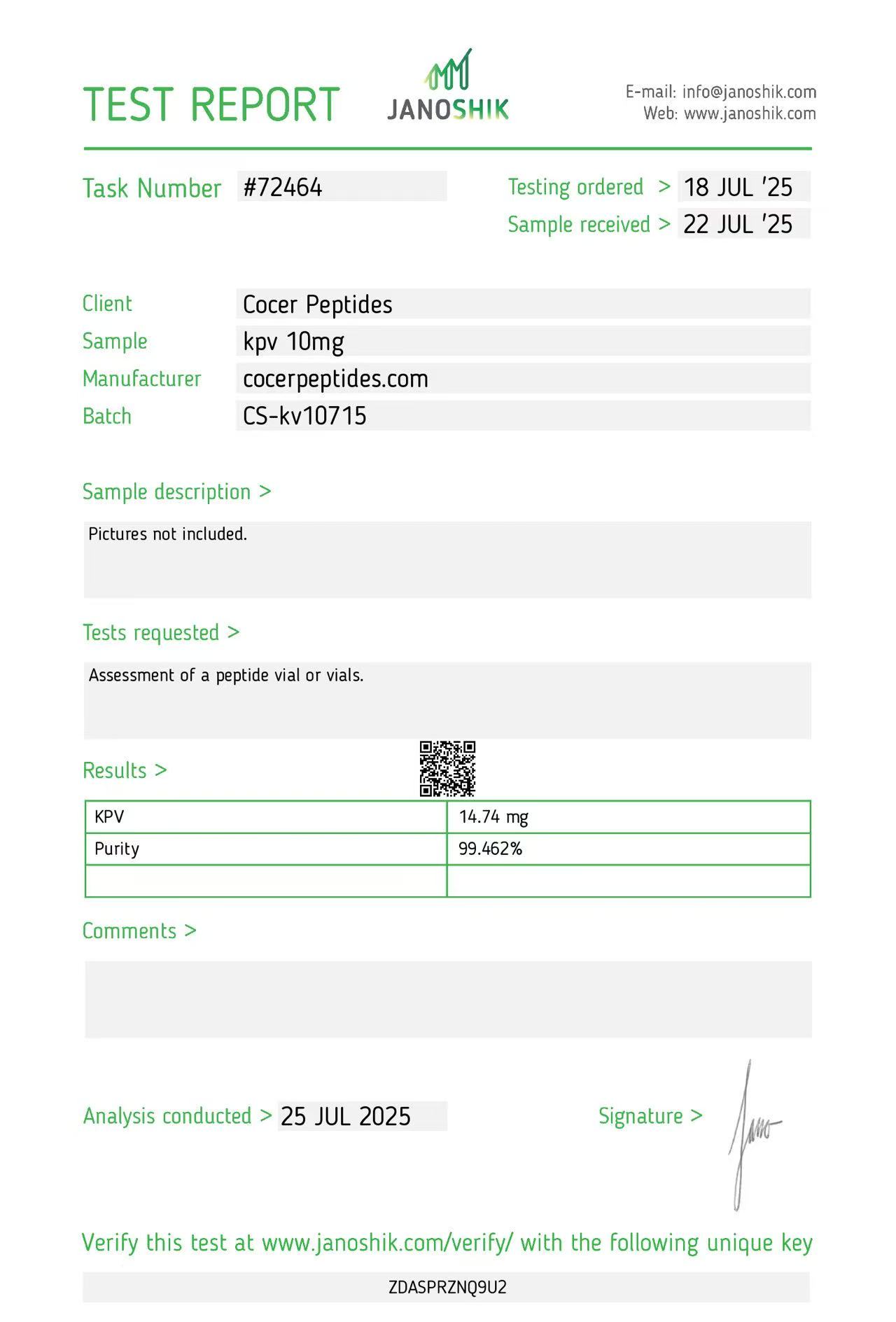 Click to open report
Click to open report
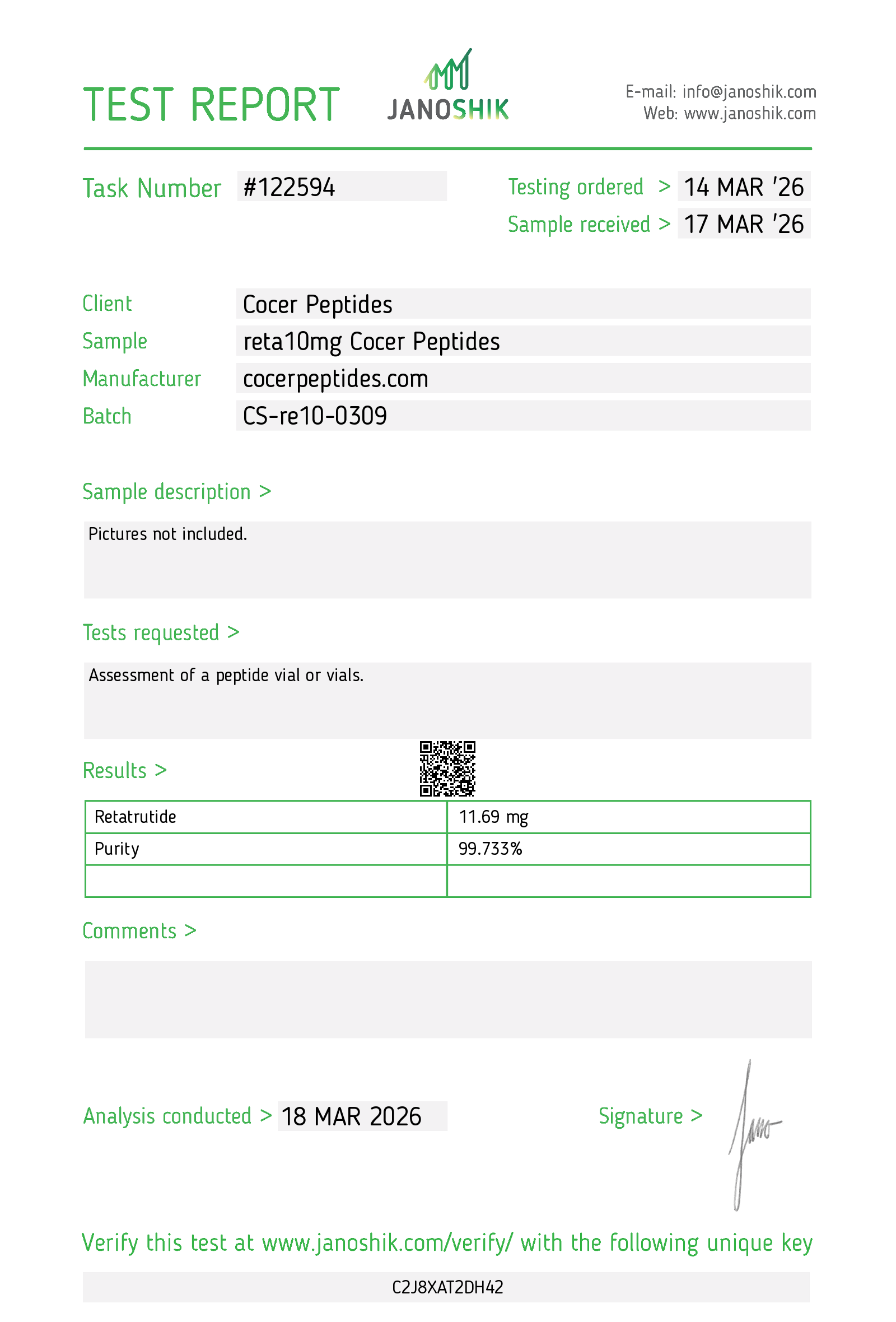
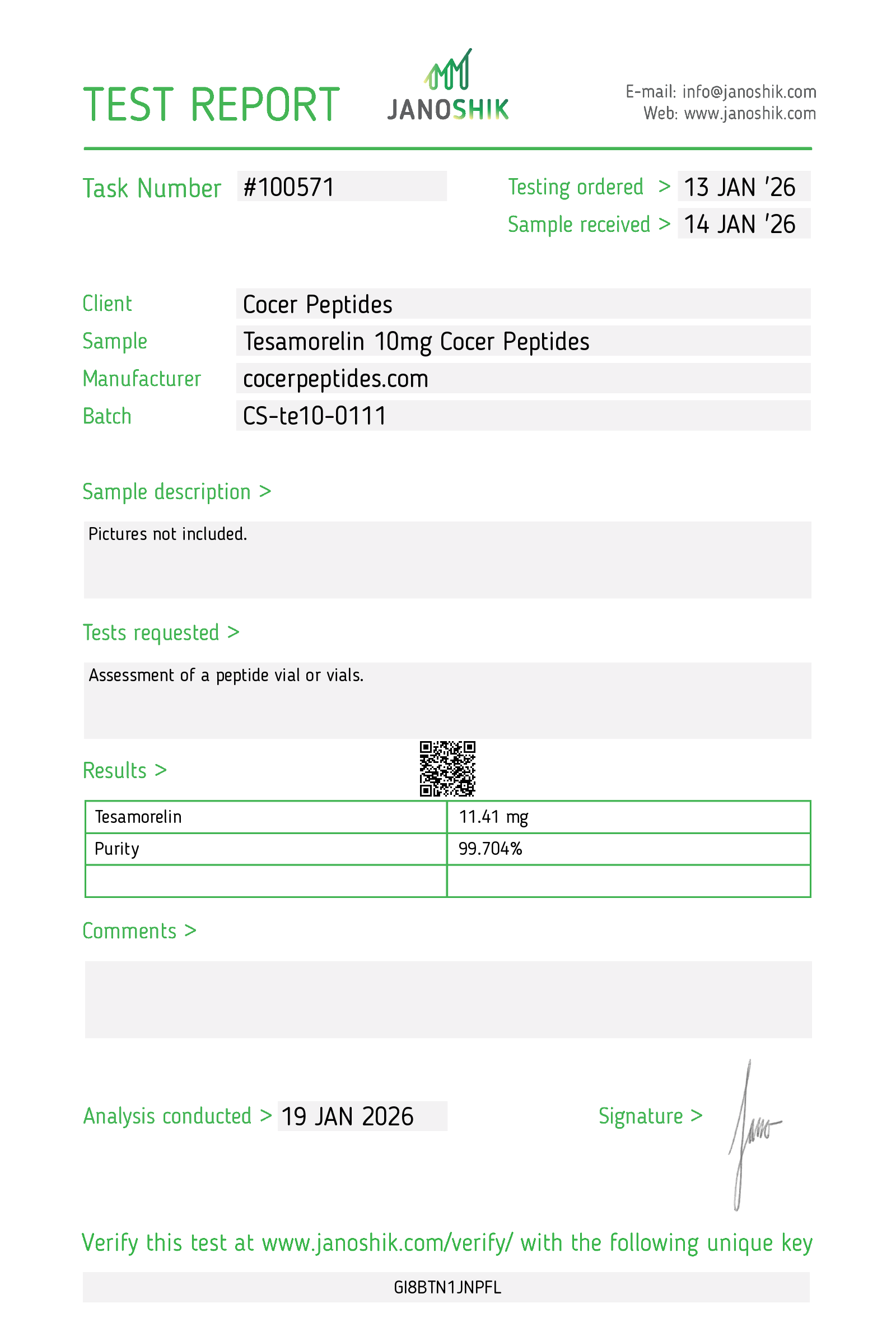
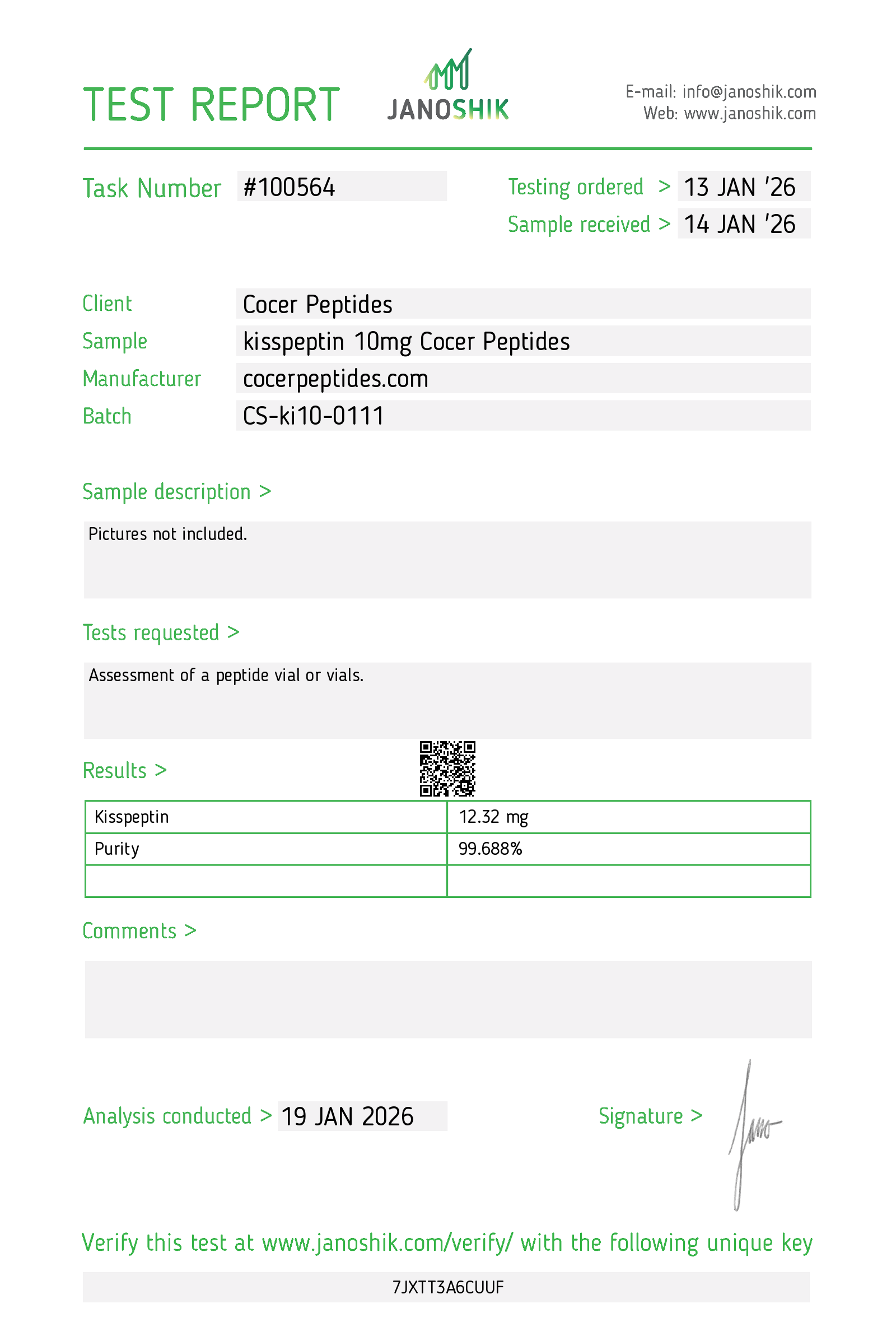
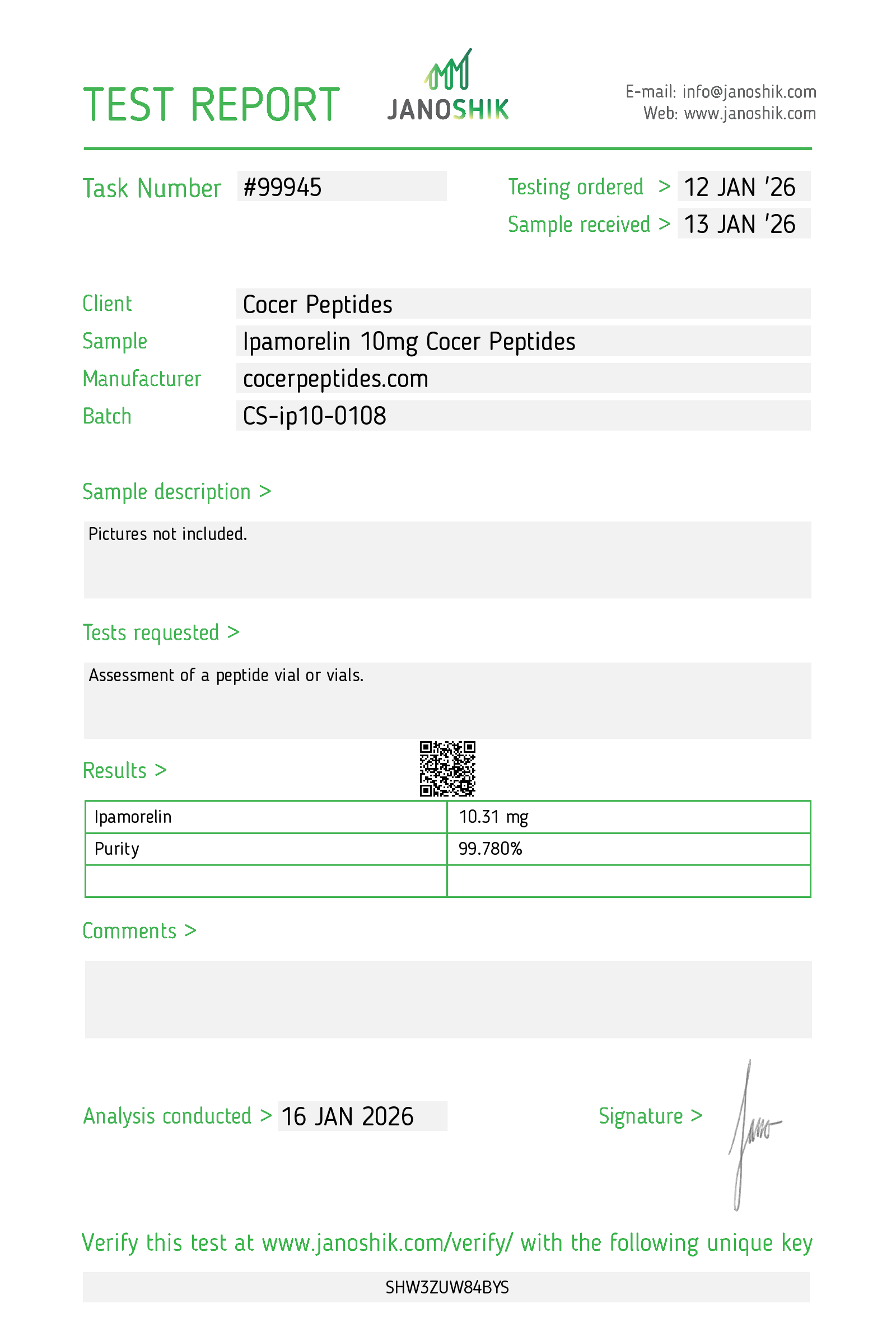
Every HGH and peptide product sold by Lumera Labs is tested by an independent third-party laboratory — Janoshik Analytical — before it ships. The only exception is pharmaceutical Bacteriostatic Water (manufactured by Pfizer). If a lot is not tested, we do not sell it.
Each product is paired with its Janoshik Certificate of Analysis. Cross-reference the batch number on your vial against the report on this page — the document is publicly viewable and verifiable.
A retain sample from every released lot is held under storage spec for five years from release date. If your assay drifts during that window, we re-run our material against yours at no charge.